COVID-19 Antigen Test Kit (FDA EUA Approved)
This COVID-19 Antigen Test Kit (FDA EUA Approved) is a rapid, accurate, and reliable test for the detection of SARS-CoV-2, the virus that causes COVID-19. This test is designed to detect the presence of the virus in the upper respiratory tract and is intended for use in individuals who are suspected of having an active COVID-19 infection.
The test is easy to use and provides results in as little as 15 minutes. The test kit includes a nasal swab, a sample collection tube, and a test cassette. The nasal swab is used to collect a sample from the upper respiratory tract, which is then placed into the sample collection tube. The sample is then added to the test cassette, which contains a reagent that reacts with the virus if it is present. The results are then read from the test cassette, with a positive result indicating the presence of the virus.
The COVID-19 Antigen Test Kit (FDA EUA Approved) is highly accurate, with a sensitivity of 97.2% and a specificity of 99.6%. It is also easy to use, with minimal training required. The test is also cost-effective, making it an ideal choice for screening large numbers of people. Overall, the COVID-19 Antigen Test Kit (FDA EUA Approved) is an accurate, reliable, and cost-effective way to detect the presence of SARS-CoV-2 in individuals who are suspected of having an active COVID-19 infection.
SKU:
CD-CO291-2 / CD-CO291-5
Categories: Clinical Diagnostics, COVID-19, Flu & RSV, Infectious Diseases
Description
The COVID-19 Ag Rapid Test does not differentiate between SARS-CoV and SARS-Cov-2.
Results are for the identification of SARS-CoV-2 nucleocapsid protein antigen, which is generally detectable in anterior nasal (nares) swab specimens during the acute phase of infection. Positive results indicate the presence of viral antigens, but clinical correlation with past medical history and other diagnostic information is necessary to determine infection status. Positive results do not rule out bacterial infection or co-infection with other viruses. The agent detected may not be the definite cause of disease. Individual who tests positive with the COVID-19 Ag Rapid Test should self-isolate and seek follow-up care with their physician or healthcare provider as additional testing may be necessary.
All negative results are presumptive and confirmation with a molecular assay, if necessary for patient management, may be performed. Negative results do not rule out SARS-CoV-2 infection and should not be used as the sole basis for treatment or patient management decisions, including infection control measures such as isolating from others and wearing masks. Negative results should be considered in in the context of an individual’s recent exposures, history, and the presence of clinical signs and symptoms consistent with COVID-19.
Individual shall provide all results obtained with this product to their healthcare provider for public health reporting and to receive appropriate medical care.
TEST PROCEDURES
Step 1: Sample Collection
• Remove the swab from the pouch (Note: Be careful not to touch the swab tip (soft end) with fingers.)
• Carefully insert the swab tip into one nostril about ½ to ¾ inch. Firmly and slowly rotate the swab 5 times, brushing against the inside walls of the nostril to ensure both mucus and cells are collected. (Note: 1. Do not push the swab further if you meet resistance. 2. For young children, do not insert more than ½ inch.)
• Using the same swab, repeat this process for the other nostril to ensure an adequate sample is collected from both nostrils. (Note: Failure to swab properly may cause incorrect results.)
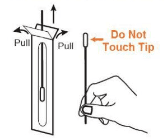
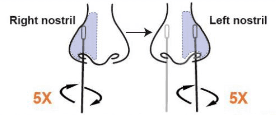
Step 2: Sample Processing
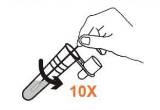
• Insert the swab in tube to the bottom.
• Rotate the swab at least 10 times while pressing the swab head against the bottom and side of the tube.
• Remove the swab while squeezing the sides of the tube to express as much liquid as possible from the swab.
• Attach the dropper tip firmly onto the tube.
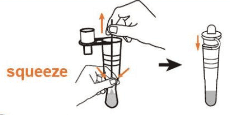
Step 3: Sample Testing
• Gently squeeze the tube and dispense 3 drops of solution into the sample well. (Note: A false negative or invalid result may occur if less than 3 drops of solution are added to the Sample well.)
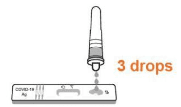
Step 4: Testing Result
• The result is valid when read at 10 – 30 minutes. If a POSITIVE result is obtained within 10 minutes, the result should also be considered valid.

Step 5: Guidelines on Test Result Interpretation
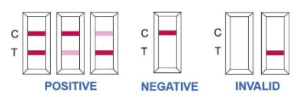
DOCUMENT FOR DOWNLOADING
>> Materials Safety Data Sheet
[wpforms id=”4979″]
Additional information
| Product No |
CD-CO291-2 ,CD-CO291-5 |
|---|---|
| Packing Size |
2 tests ,5 tests |
Shipping & Delivery
Related products
0.65 ml Safeseal MCT – Microcentrifuge Tubes
$199.99
Select options
This product has multiple variants. The options may be chosen on the product page
55 ml Reagent Reservoir
$199.99
Select options
This product has multiple variants. The options may be chosen on the product page
Marijuana (THC) Drugs Test
$25.99 – $109.99Price range: $25.99 through $109.99
Select options
This product has multiple variants. The options may be chosen on the product page
Microcentrifuge Tube Rack
$99.99
Select options
This product has multiple variants. The options may be chosen on the product page